Carbon capture in an electrochemical power cell
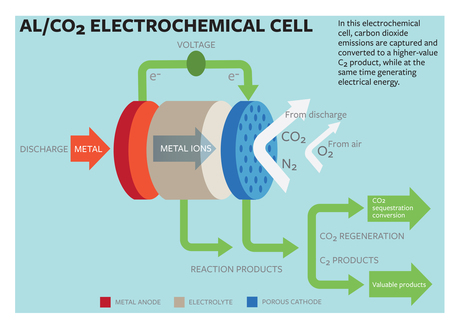
Cornell University researchers have developed a method for capturing carbon dioxide, converting it into a useful product and producing electrical energy. Writing in the journal Scientific Advances, Professor Lynden Archer and doctoral student Wajdi Al Sadat describe an oxygen-assisted power cell that uses electrochemical reactions to both sequester the CO2 and produce the electricity.
In most current carbon-capture models, the carbon is captured in fluids or solids, which are then heated or depressurised in energy power plants to release the CO2. As noted by Professor Archer, “The regeneration of the fluids used for capturing carbon dioxide utilise as much as 25% of the energy output of the plant. This seriously limits commercial viability of such technology.”
Furthermore, the concentrated gas must then be compressed and either transported to industries able to re-use it or sequestered underground. This requires new infrastructure, Professor Archer added.
As an alternative, the Cornell scientists have proposed a power cell that uses aluminium as the anode and mixed streams of CO2 and oxygen as the active ingredients of the cathode. The electrochemical reactions between the anode and the cathode sequester the CO2 into carbon-rich compounds while also producing electricity and a valuable oxalate as a by-product.
“The fact that we’ve designed a carbon capture technology that also generates electricity is, in and of itself, important,” Professor Archer said, with the electrochemical cell generating 13 ampere hours per gram of porous carbon (as the cathode) at a discharge potential of around 1.4 volts. The energy produced by the cell is said to be comparable to that produced by the highest energy-density battery systems.
Another key aspect of the team’s findings is in the generation of superoxide intermediates, which are formed when the dioxide is reduced at the cathode. The superoxide reacts with the normally inert carbon dioxide, forming a carbon-carbon oxalate that is widely used in many industries, including pharmaceutical, fibre and metal smelting.
“A process able to convert carbon dioxide into a more reactive molecule such as an oxalate that contains two carbons opens up a cascade of reaction processes that can be used to synthesise a variety of products,” said Professor Archer, noting that the configuration of the electrochemical cell will be dependent on the product one chooses to make from the oxalate.
Al Sadat said aluminium is the perfect anode for this cell, as it is plentiful, safer than other high-energy density metals and lower in cost than other potential materials (lithium, sodium) while having comparable energy density to lithium. He added that many aluminium plants are already incorporating some sort of power-generation facility into their operations, so this technology could assist in both power generation and reducing carbon emissions.
“It [also] fits really well with onboard capture in vehicles, especially if you think of an internal combustion engine and an auxiliary system that relies on electrical power,” he noted.
A current drawback of this technology is that the electrolyte — the liquid connecting the anode to the cathode — is extremely sensitive to water. Ongoing work is addressing the performance of electrochemical systems and the use of electrolytes that are less water-sensitive.
Battery study explores safer rapid-charging methods
Scientists from Adelaide University have demonstrated a fast-charging battery approach designed...
Nanoscale device converts wasted light into energy
UNSW Sydney scientists have addressed a longstanding problem in photonics: how to stop energy...
Monash researchers break barrier in membrane design
Engineers have developed an ultra-thin membrane that allows fuel cells to operate more...




